Ex.18 In the system, LaCl3(s) + H2O(g) + Heat = LaCIO(s) + 2HCl(g), is established. More water vapour is added to reestablish the equilibrium. The pressure of water vapour is doubled. The

Click here:point_up_2:to get an answer to your question :writing_hand:ex18in the systemlacl3s h2og heat lacios 2hclgis established more water vapour is
Click here👆to get an answer to your question ✍️ Ex-18 In the system- LaCl3-s- - H2O-g- - Heat - LaCIO-s- - 2HCl-g- is established- More water vapour is added to reestablish the equilibrium- The pressure of water vapour is doubled- The factor by which pressure of HCl is changed is - -A-2 -B- V -C- 13 -D
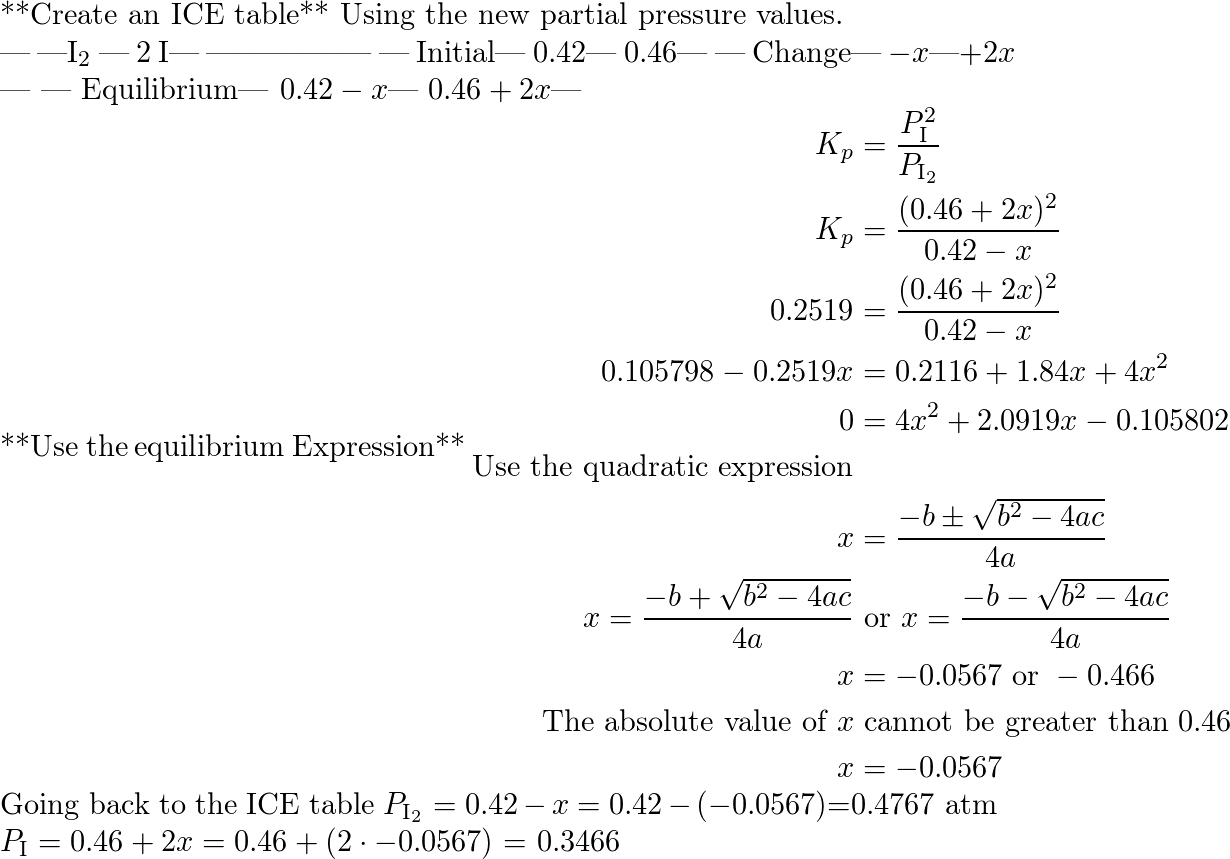
A system at equilibrium contain s $l_2(g)$ at a pressure of

Chemistry 2 Unit 11 Flashcards

Chemistry 2 Flashcards

Solved For the system water (1), ethyl acetate (2), and

CH3-The Second and Third Laws of Thermodynamics.pdf - CHAPTER 3 The Second and Third Laws of Thermodynamics LAIDLER . MEISER . SANCTUARY Physical

Question Video: Determining Which Solution Will Have the Lowest Vapor Pressure


Ex.18 In the system, LaCl3(s) + H2O(g) + Heat = LaCIO(s) + 2HCl(g), is established. More water vapour is added to reestablish the equilibrium. The pressure of water vapour is doubled. The
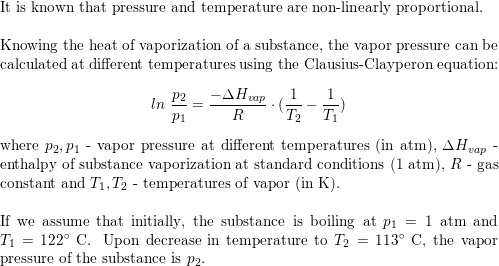

To the system, LaCl(3)(s)+H(2)O(g) hArr LaClO(s)+2HCL(g)-Heat alre

Thermochemistry and Thermodynamics - ppt download

Consider this reaction at equilibrium: C(s) + H2O(g) ⇌ CO(g) + H2

Thermochemistry and Thermodynamics - ppt download
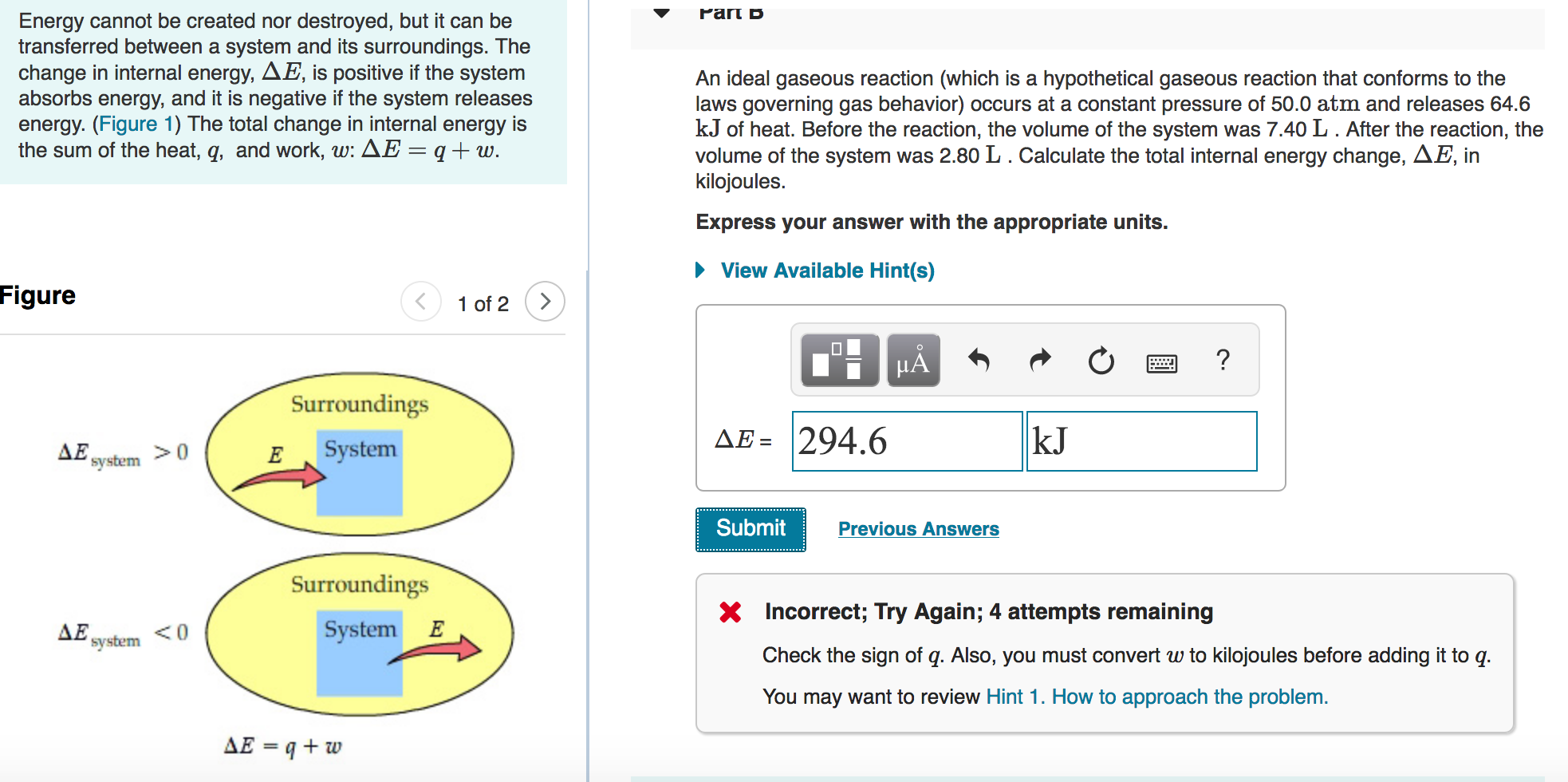
Solved b. An ideal gaseous reaction (which is a hypothetical









