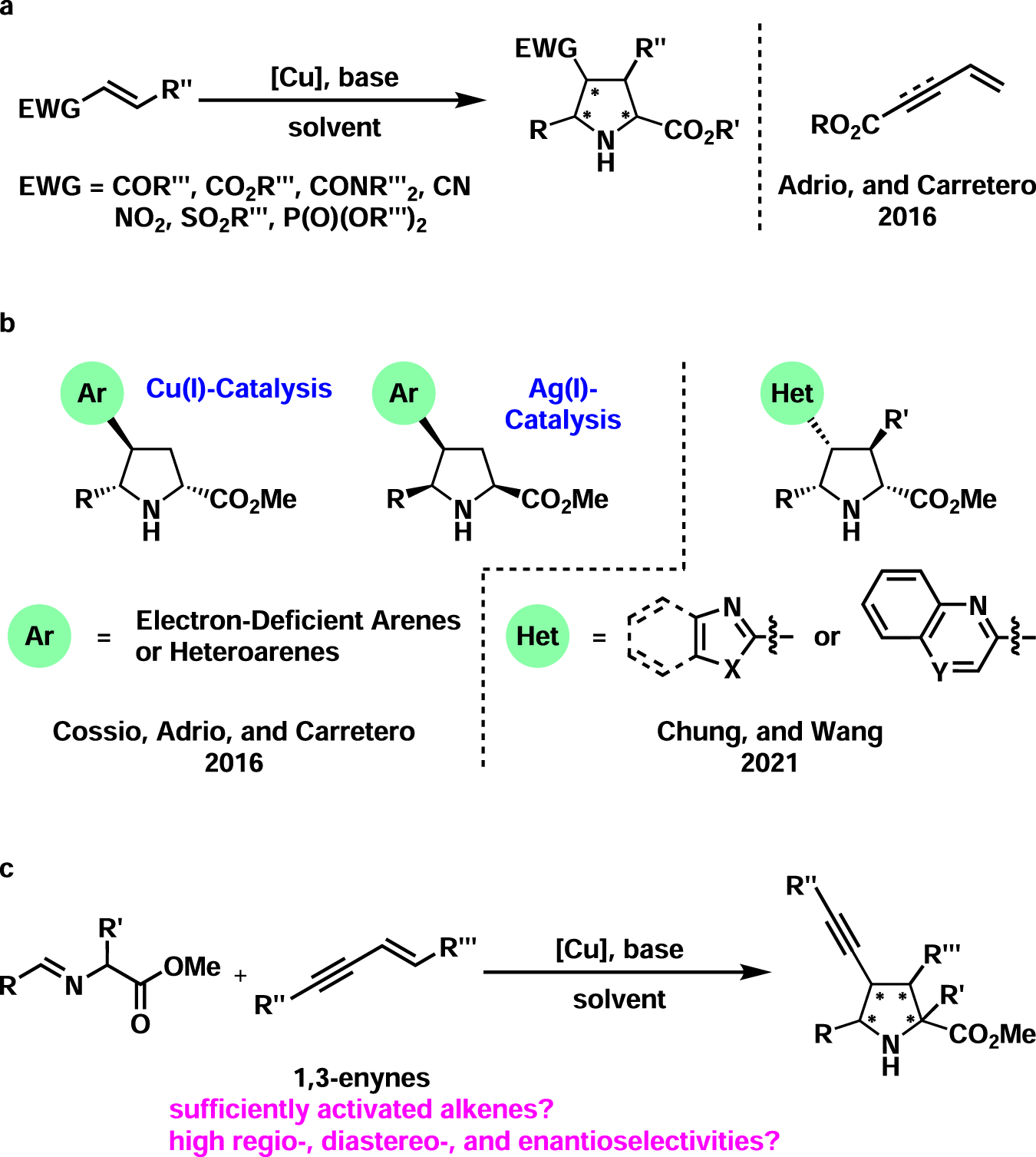
Copper(I)-catalyzed asymmetric 1,3-dipolar cycloaddition of 1,3-enynes and azomethine ylides

PDF] Halogenated salt assisted Cu-catalyzed asymmetric 1,4-borylstannation of 1,3-enynes: enantioselective synthesis of allenylstannes

Copper(I)-Catalyzed Asymmetric 1,3-Dipolar Cycloaddition of Azomethine Ylides with Fluorinated Imines: The Expanded Scope and Mechanism Insights
Copper-catalyzed functionalization of enynes

Kinetic Resolution of Alkylidene Norcamphors via a Ligand-Controlled Umpolung-Type 1,3-Dipolar Cycloaddition. - Abstract - Europe PMC

Ligand-controlled stereodivergent 1,3-dipolar cycloaddition of azomethine ylides with 3-methyl-4-nitro-5-styrylisoxazoles.

Strategies for synthesis of chiral exocyclic allenes. a-c Previous
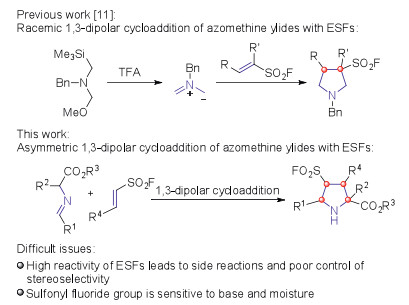
Cu-catalyzed endo-selective asymmetric 1,3-dipolar cycloaddition of azomethine ylides with ethenesulfonyl fluorides: Efficient access to chiral pyrrolidine-3-sulfonyl fluorides

Copper(I)-catalyzed asymmetric 1,3-dipolar cycloaddition of 1,3-enynes and azomethine ylides

Copper-catalyzed functionalization of enynes

Copper(I)-catalyzed asymmetric 1,3-dipolar cycloaddition of 1,3-enynes and azomethine ylides

Kinetic Resolution of Alkylidene Norcamphors via a Ligand-Controlled Umpolung-Type 1,3-Dipolar Cycloaddition. - Abstract - Europe PMC

Copper-catalyzed enantioselective hydrofunctionalization of 1,3-enynes.

Copper(I)-catalyzed asymmetric 1,3-dipolar cycloaddition of 1,3-enynes and azomethine ylides

Cu-catalyzed asymmetric 1,4-functionalization of 1,3-enynes

Cu-catalyzed endo-selective asymmetric 1,3-dipolar cycloaddition of azomethine ylides with ethenesulfonyl fluorides: Efficient access to chiral pyrrolidine-3-sulfonyl fluorides - ScienceDirect
/oreillers-de-corps-sourire-et-des-dents-zebre.jpg.jpg)