PDF) A Multicenter Phase II Trial of Docetaxel, Cisplatin, and Cetuximab ( TPEx) Followed by Cetuximab and Concurrent Radiotherapy for Patients With Local Advanced Squamous Cell Carcinoma of the Head and Neck (CSPOR

A Multicenter Phase II Trial of Docetaxel, Cisplatin, and Cetuximab (TPEx) Followed by Cetuximab and Concurrent Radiotherapy for Patients With Local Advanced Squamous Cell Carcinoma of the Head and Neck (CSPOR HN01: ECRIPS Study)

Radiotherapy plus cetuximab or cisplatin in head and neck squamous cell carcinoma: an updated systematic review and meta-analysis of randomized controlled trials

Efficacy and toxicity of weekly paclitaxel, carboplatin, and cetuximab as induction chemotherapy or in cases of metastases or relapse for head and neck cancer with a focus on elderly or frail patients

A randomized phase III trial (RTOG 0522) of concurrent accelerated radiation plus cisplatin with or without cetuximab for stage III-IV head and neck squamous. - ppt download

Head and Neck Cancers, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology in: Journal of the National Comprehensive Cancer Network Volume 18 Issue 7 (2020)
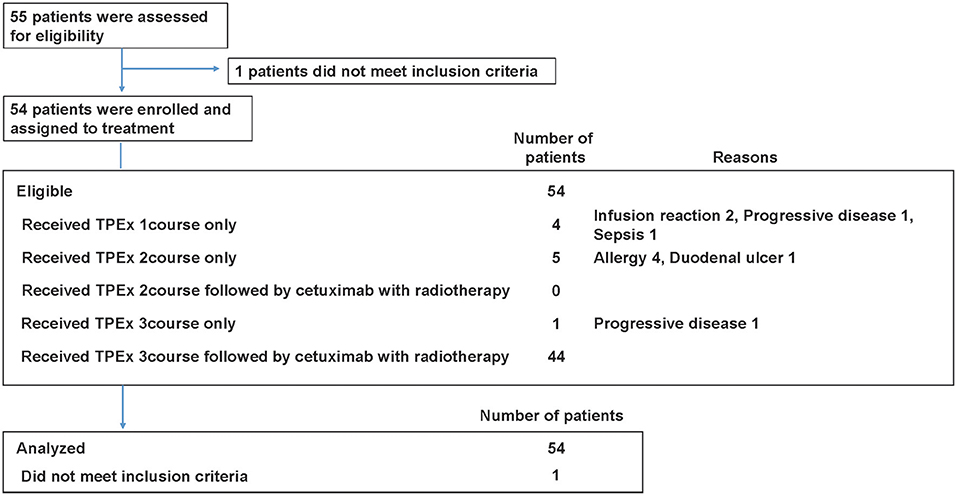
Frontiers A Multicenter Phase II Trial of Docetaxel, Cisplatin, and Cetuximab (TPEx) Followed by Cetuximab and Concurrent Radiotherapy for Patients With Local Advanced Squamous Cell Carcinoma of the Head and Neck (
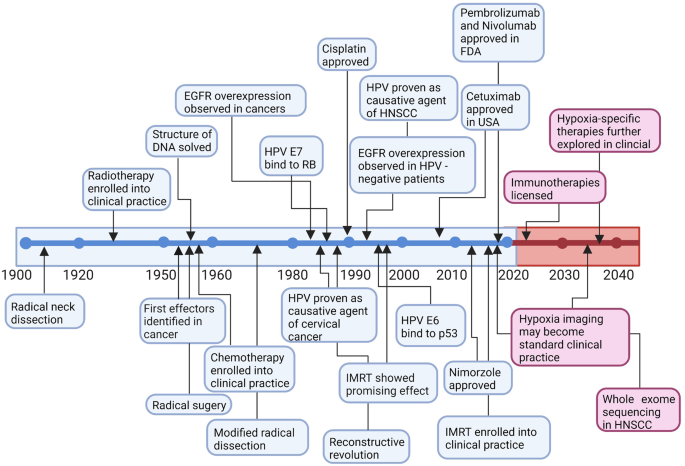
Targeted therapy for head and neck cancer: signaling pathways and clinical studies
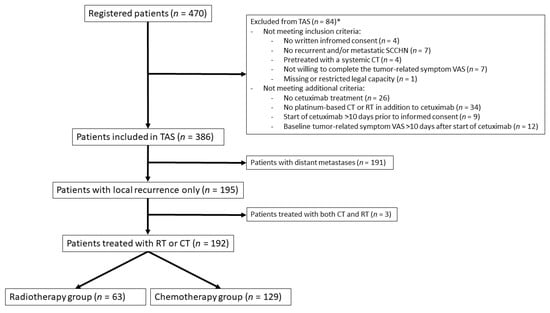
Cancers, Free Full-Text

PDF) A Randomized, Multicenter, Phase II Study of Cetuximab With Docetaxel and Cisplatin as Induction Chemotherapy in Unresectable, Locally Advanced Head and Neck Cancer

PDF) A Multicenter Phase II Trial of Docetaxel, Cisplatin, and Cetuximab ( TPEx) Followed by Cetuximab and Concurrent Radiotherapy for Patients With Local Advanced Squamous Cell Carcinoma of the Head and Neck (CSPOR









